February 26, 2026
Layers of Brilliance - Episode Four: The Breakthrough


February 26, 2026
Layers of Brilliance - Episode Four: The Breakthrough
When Katharine Burr Blodgett discovers non-reflecting glass, The General Electric Company’s public relations machine makes her a star.

Episode Description
The 1930s prove to be an exceptional decade for research at The General Electric Company. Katharine Burr Blodgett works closely alongside her boss, Irving Langmuir who, in 1932, wins the Nobel Prize for Chemistry. In 1938, Katharine’s meticulous experiments with thin film coatings on solid surfaces lead to her most important breakthrough: non-reflecting glass.
The General Electric Company’s public relations machine kicks into high gear. Katharine becomes an overnight sensation, both in the scientific community and in the press, which dub her discovery “invisible glass.” The assistant to the Nobel Prize winner, long invisible herself, takes center stage.


Katie is co-founder and co-executive producer of The Lost Women of Science Initiative. She is the author of six nonfiction books and one novel, and was a longtime reporter for The New York Times. She is at work on her second novel.

Natalia is a Peruvian journalist, editor, and writer based in Philadelphia. Her work focuses on gender inequality, labor issues, and reproductive rights. Natalia has worked as an editor for Radio Ambulante at NPR and in 2021, she won the Aura Estrada International Literary Award. She is currently working on her first book.

Sophia Levin is a journalist and teacher based in Washington, D.C. and Pittsburgh, PA. She has written about unions, infrastructure, and reproductive healthcare for The Tartan and PublicSource. She holds a bachelor’s degree in Creative Writing and History from Carnegie Mellon University.

Hannah is a Boston-based journalist with a background in storytelling, production, and strategic communication. She has reported on a variety of topics, including arts, culture, finance, and relationships between municipal agencies and communities. She studied journalism and art history at Northeastern University.

Katie is co-founder and co-executive producer of The Lost Women of Science Initiative. She is the author of six nonfiction books and one novel, and was a longtime reporter for The New York Times. She is at work on her second novel.

Natalia is a Peruvian journalist, editor, and writer based in Philadelphia. Her work focuses on gender inequality, labor issues, and reproductive rights. Natalia has worked as an editor for Radio Ambulante at NPR and in 2021, she won the Aura Estrada International Literary Award. She is currently working on her first book.

Sophia Levin is a journalist and teacher based in Washington, D.C. and Pittsburgh, PA. She has written about unions, infrastructure, and reproductive healthcare for The Tartan and PublicSource. She holds a bachelor’s degree in Creative Writing and History from Carnegie Mellon University.

Hannah is a Boston-based journalist with a background in storytelling, production, and strategic communication. She has reported on a variety of topics, including arts, culture, finance, and relationships between municipal agencies and communities. She studied journalism and art history at Northeastern University.
Michael Petty is a retired engineering professor at Durham University in England and author of the 2008 book, Langmuir-Blodgett Films: An Introduction.
Eric Furst is the William H. Severns Jr. Distinguished Chair of Chemical Engineering at the University of Delaware.
Peggy Schott is a retired chemist from Northwestern University and has written about Katharine Burr Blodgett and her achievements.
Ginger Strand is an American author of nonfiction and fiction. She is the author of the 2015 nonfiction book, The Brothers Vonnegut: Science and Fiction in the House of Magic.


Further Reading:
Langmuir-Blodgett Films: An Introduction, by Michael C. Petty, Cambridge University Press, 2008.
Electrical Processes in Organic Thin Film Devices: From Bulk Materials to Nanoscale Architectures, by Michael C. Petty, John Wiley & Sons, Inc., 2022.
Langmuir-Blodgett Films: Fundamentals to Applications, by Syed Arshad Hussain, Elsevier Science, 2025.
The Brothers Vonnegut: Science and Fiction in the House of Magic, by Ginger Strand, Farrar, Straus and Giroux, 2015.
The Remarkable Life and Work of Katharine Burr Blodgett (1898–1979), by Margaret E. Schott, E. Thomas Strom, and Vera V. Mainz from In The Posthumous Nobel Prize in Chemistry Volume 2, Ladies in Waiting for the Nobel Prize 1311, 1311:151–82. Washington, DC: American Chemical Society, 2018.
Episode Transcript
Episode 4 – The Breakthrough
Katie Hafner: This is our fourth episode in our season about Katharine Burr Blodgett. If you haven't heard the other episodes, please do go back to the first one and start there.
At 1:00 p.m. on November 10th, 1932, Irving Langmuir got a phone call. It was a reporter calling from Sweden, who said he’d heard a rumor that Langmuir was to be awarded the Nobel Prize in Chemistry.
Langmuir was cautious. He’d heard nothing official. But within hours the rumor became fact. The prize was awarded for the work Langmuir had done sixteen years earlier on molecular monolayers, work that essentially invented the field of surface chemistry.
A month later, Langmuir was in Stockholm with his wife and children to accept the prize.
There was a whirlwind of pomp and ceremony. Banquets. Speeches. The medal.
By then, Irving Langmuir was already a familiar figure inside the Nobel system. He’d been nominated again and again since 1916 — five times in physics and nine times in chemistry — by the same small circle of senior men who dominated science at the time. On his tenth chemistry nomination, the Nobel committee finally said yes .
It was an old boys’ network in the most literal sense: professors nominating professors, Nobel laureates nominating future laureates, the same names circulating year after year. Langmuir belonged to that system. And the 1932 prize was Langmuir's moment - the system’s recognition of one of its own and the public affirmation of a man who had always believed in his own brilliance.
After that, Langmuir’s career entered a different orbit. Still, he was a scientist through and through. And when he returned to Schenectady, he had already decided what to pursue next… or rather, what he would go back to: molecular monolayers.
The Nobel Prize had cast a clarifying light backward. Monolayers were something he had set aside in favor of flashier problems, including, as we saw last week, his failed attempt to describe the inner workings of atoms.
Now monolayers stood at the center of his reputation -- and they had his full attention. As soon as he got back to the lab, Langmuir suggested to Katharine Blodgett that they circle back to monolayers.
And for Katharine, that decision would shape the most important science of her career.
I’m Katie Hafner and this is Lost Women of Science. Today on Layers of Brilliance, what happens when the humble assistant becomes the star?
The idea that oil spreads across water is ancient.
Fishermen knew it.
Sailors knew it.
So did Benjamin Franklin, who in 1774 reported that he poured a teaspoon of oil onto a pond in England and watched it spread -- smooth as a looking glass, shimmering with color.
What Franklin didn’t know—but what scientists would later begin to suspect—was just how thin that film really was.
Here's Michael Petty, an engineering professor at the University of Durham in England and author of a book on Langmuir and Blodgett's work in material science.
Michael Petty: If you look at, uh, the paper, uh, that, uh, Benjamin Franklin wrote describing his experiment, he says he took a teaspoon of oil and it spread out into an area of, uh, about a half an acre, something like that.
And if you actually do the sums,
Katie Hafner: Which is what Irving Langmuir did in 1916…
Michael Petty: You can work out that his film was probably about a molecule in thickness.
Katie Hafner: Langmuir found ways not only to measure that layer, but to compress it, to study how it behaved under controlled conditions — which was part of the research that earned him the Nobel Prize.
Langmuir also showed that when oil spreads across water, it doesn’t thin out endlessly. The molecules spread out onto a surface to form a film just one molecule thick. Then they stop. And sit there. This process in which molecules stick to a surface rather than sinking into it, called adsorption, is as you might remember, the same one Katharine studied in her graduate work at the University of Chicago and continued to play with when she arrived at GE.
Back when Katharine and Langmuir first worked on monolayers—with Katharine running most of the experiments, of course—they'd studied not just the properties of monolayers, but how to manipulate them. Like how to transfer a monolayer of oil resting on the surface of water, to a metal plate.
All of this work came before the Nobel Prize.
So when Langmuir returned home in the summer of 1933, the question before them was: What else can we do with these layers?
Katharine got to work. She was using a metal trough that was invented in 1882 by Agnes Pockels, a self-trained German chemist. Pockels developed her trough to study what happens when substances interact on the surface of water. (We’re going to devote a whole episode to Agnes in March, by the way.)
Using a modified version of Pockels’ trough, Katharine dipped solid plates in and out of the water, observing the water’s surface tension and the light reflecting from the plates. She concocted every possible type of problem to explore surface chemistry, attempting to manipulate the monolayer as it transferred from water to the solid surface.
What could she do with just one monolayer, one molecule thick, other than examine how it behaved on the surface of water? She was stuck.
Time and again, for about six months, Katharine was left with her invisible film staring back at her. What wasn’t she seeing?
These were not experiments you could rush. They were unforgiving. They demanded steadiness, repetition, and a tolerance for failure that few people possessed. The films Katharine was working with were almost impossibly delicate.
Over the years, when people who worked with Katharine Blodgett or knew her work described her, they didn’t reach first for words like brilliant or visionary.
They talked instead about HOW she worked.
About what it took to do the kind of science she did.
And there was one word that cropped up over and over again:
Michael Petty: Katharine Blodgett was a very, very patient scientist,
Vincent Schaefer: In addition to her patience, of course,
Eric Furst: She just had tremendous patience
Peggy Schott: Patience
Guy Suits: Persistence and patience
Vincent Schaefer: One of the most important things I learned from Katie was patience.”
Katie Hafner: Katharine paid attention to the smallest details, not because she was fussy – okay, maybe she was a little fussy – but because she knew that at the scale she worked on, everything mattered.
But it’s at this point that we encountered a problem. And it’s not a scientific one – it’s a historical one. So far, we don’t have Katharine Blodgett’s laboratory notebooks from this period where she would have detailed the results of each experiment. We have Irving Langmuir’s notebooks, of course, and a handful of recollections from colleagues. And we have a brief description by Katharine herself, years after the fact.
But what’s missing, most of all, is the real-time record of her hands at work.
So the story has to be pieced together. It’s an act of reconstruction – necessarily incomplete. In effect, we have to imagine the endlessly repetitive experiments that led to the discoveries.
Here’s Katharine’s co-worker of many years, Vincent Schaefer, talking about the process of doing science.
Vincent Schaefer: What science is all about and how you go about doing it. And it is so different than the conception that most young people have. A lot of them think that you set out a problem. And you head toward it and you solve it. And that isn't the way it works at all. You blunder along, and you don't head for the objective. You go this way and you go that way. And very frequently a serendipitous event occurs and you have something much more important than the thing you're looking for
Katie Hafner: Indeed, in an interview after her retirement, Katharine described her research process overall. She said, “You keep barking up so many wrong trees in research. It seems sometimes as if you’re going to spend your whole life barking up wrong trees. And I think there is an element of luck if you happen to bark up the right one.”
And with luck, genius, and relentless patience, eventually, Katharine found the right tree to bark up. Dipping her metal plate in and out of a water trough, months after she began this set of experiments, she noticed something new. Here’s Peggy Schott.
Peggy Schott: In December, 1933 when Katharine one day was working in the laboratory, she's using talc
Katie Hafner: That’s ordinary talcum powder, which she used for a very simple, but important reason: the films themselves were so thin as to be invisible.
Peggy Schott: She needs a visual aid to see movement, like physical movement of the layers as they're going onto the plate.
Katie Hafner: In other words, she needed a way to see motion without needing to see the molecules themselves. If you sprinkle a very fine powder like talcum powder onto the surface, something magical happens:
The talc grains don’t dissolve. They float on the water. And they get pushed around by changes in surface tension.
So when a molecular film spreads, compresses, or gets transferred onto a solid plate, it changes the surface tension of the water. The talc grains respond to that – they move.
In other words: The talc turns invisible molecular motion into visible patterns.
It’s like smoke in the air, revealing how the air is flowing.
The talc was Katharine’s visual tracer.
Peggy Schott: So she sprinkles talcum powder on top of the oil.
Katie Hafner: And she lowers the plate…
Peggy Schott: The plate’s going down into the trough full of liquid.
Peggy Schott: And, as she lowers the plate, the invisible layer coats the plate, and the talc moves toward the plate.
Katie Hafner: And that's her visual indicator of what's happening at the molecular level. The one-molecule thick layer of oil is coating the plate and...
Peggy Schott: She can see that that’s happening. She has the metal plate, uh, down into the water, and now she's going to raise it up.
Katie Hafner: Katharine expected that the talc would remain stationary, indicating that just a single layer of molecules adhered to the plate. … So she dips it into the water again. But as she raises it…
Peggy Schott: … The surprise was that the talc moved again, indicating that another layer of oil went onto the first layer on the plate.
Katie Hafner: So she could tell that she was creating multiple layers - each monolayer one molecule thick.
Peggy Schott: That was her eureka moment, that's her discovery.
Katie Hafner: Vincent Schaefer was there in the lab that night, too. And years later, he recalled the moment.
Vincent Schaefer: I saw Katie dip the very first buildup film. In fact, I didn't quite believe it and I, when she wasn't looking, I tried it myself and it worked.
Katie Hafner: Katharine could now stack layers of a substance on top of a solid surface. It would take about 40,000 of these films stacked on top of each other to get to the thickness of one sheet of paper.
GE knew they had something important here. Today any surface that has a coating, any device that uses stacks of extremely thin films — television screens, cell phone screens and the microchips inside — can be traced back to Katharine, repeatedly dipping a metal plate in a trough of water.
Katharine took this research and she ran with it. She published the results from that work in 1935 and over the years GE applied for a series of patents for this method of creating multi-layer films and its possible applications. In 15 years, GE was granted eight patents with Katharine’s name on them. She was listed as the sole inventor on six and as a co-inventor with Vincent Schaeffer on the other two.
Once she solidified a way to stack monolayers on top of each other, the lab truly became a playground of discovery. Here, again, her patience and perseverance paid off.
Looking at a tendril of soap snaking across water in your kitchen sink, or a streak of car exhaust stretching across a puddle, you may have noticed a rainbow of colors dancing over the water’s surface—irridescent shades curling into each other, reflecting light on a liquid surface.
In their research on thin films, Blodgett and Langmuir used this phenomenon to their advantage. They studied the way molecules arranged themselves on liquid surfaces by covering water with oily and soapy substances. Lipids.
When lipids sit on liquid surfaces, they reflect white light. As Katharine ran her thin film experiments, she noticed that the colors her thin films reflected weren’t random.
As she added layer after layer to a metal plate, she noticed the light reflecting off its surface change from yellow-brown to dark blue to light blue, spanning all the way to a red-yellow. She noticed that the color the thin film reflected corresponded with the number of layers it contained, and she created a color gauge to track it. For example, if the color reflected was a yellow, it corresponded to a film six-millionths of an inch thick; for red it was seven-millionths.
Rather than using expensive optical instruments to measure a film’s thickness, researchers could now use a film’s color to figure out how thick it was.
The importance of counting layers led Katharine to her most famous discovery.
MORE AFTER THE BREAK
In December 1938, Katharine Blodgett was again counting the layers that formed as she dipped a glass plate into the trough, this time containing Barium stearate, a chemical compound formed from barium and stearic acid. Each layer was just one molecule thick, on the surface, invisible to the naked eye, so there was nothing to see but the light reflecting off the plate. By then she had worked out the number of layers of barium stearate to generate specific colors based on how they reflected light. She added one layer, then another, and another… And one day, when she was doing this….
Peggy Schott: When she got to 44 layers ...
Katie Hafner: She stopped. And then…
Peggy Schott: Here’s Blodgett’s account of what happened. After dipping the slide into a tank and watching the molecular layers build up, I picked up the slide and accidentally held it to the window, and to my surprise, saw that the coated strip transmitted more light than the uncoated portions.
I sat down. Got up, walked around, thinking it over. The more I thought about it, the plainer it became, of course, that's exactly what should happen.
Katie Hafner: The layers created a non-reflective coating on the glass. Here again is her fellow GE researcher, Vincent Schaefer, recalling that moment at Katharine’s 1963 retirement dinner:
Vincent Schaefer: I was there when she very excitedly said, I can't see it. It was the non-reflecting film.
Katie Hafner: GE understood this immediately. Non-reflecting glass or invisible glass as the press started to call it, had the potential for wide applications. It could increase visibility on eyeglasses, camera lenses, and mirrors. It had the potential to protect drivers’ eyes from a windshield’s glare or increase the amount of light cameras and telescopes can take in. And museum displays! The possibilities seemed endless.
Not surprisingly, the General Electric Company's Press Office kicked into gear. And, as we’ve seen before with Langmuir, many of these stories followed a similar pattern.
Ginger Strand: If you were an editor at a newspaper, GE would present you with a full package.
That's Ginger Strand again, who's written about the history of GE.
Ginger Strand: It would be a story that was already written. It would have the photographs attached. So if you were a lazy editor, you could just take this and put it and it would be written like a news story. Even though it was PR.
Katie Hafner: What was surprising this time was the prominence they gave to Katharine herself – likely the first time the company had done something like this for a female scientist. The three-page press release, which went out in late December 1938, immediately identified Dr. Blodgett as the inventor, and quoted her extensively on what she had achieved.
That GE PR team knew what they were doing. The press was all over the story.
We sorted through hundreds of clippings in archives at newspapers.com, in scrapbooks, and file folders, each article lauding her achievements.
The New York Times covered the discovery on December 27, with a photograph of Katharine using the trough. On the same day, The New York Daily News ran the headline: “Her Discovery Makes All Glass Invisible”. And newspapers across the country picked up a report from the Associated Press wire service.
In the months that followed, Katharine was in great demand. Newspapers interviewed her, magazines profiled her. Life Magazine, for instance, ran a two-page spread.
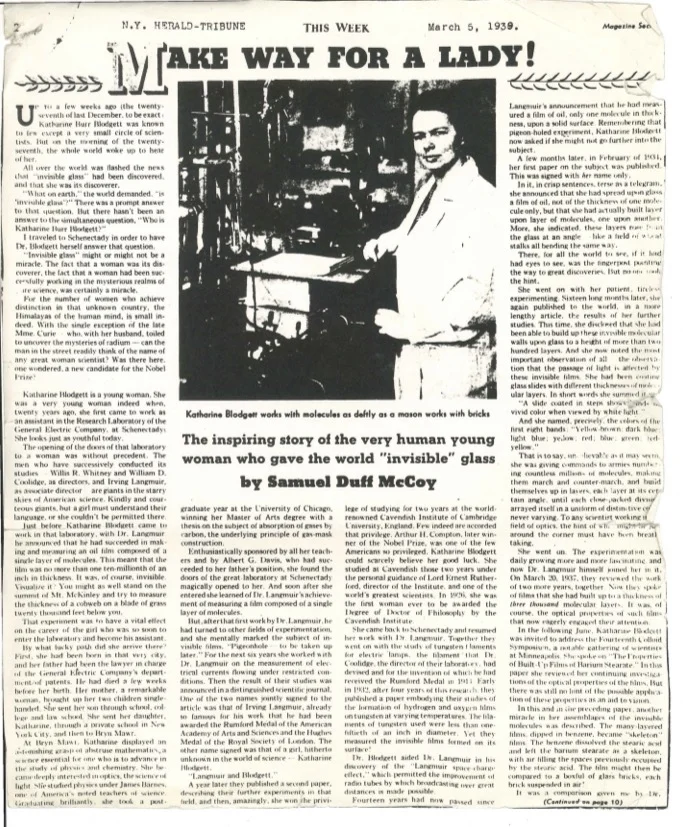
Numerous societies both social and scientific invited her to speak. The headline writers had a field day. “Magician Makes Glass Invisible,” and “It’s Witchcraft!” and “Formula for Invisible Glass is Found by Woman Scientist”, and I love this one -“The Invisible Glass Lady Throws No Stones.” In March 1939, a journalist traveled to Schenectady to watch Katharine dip the glass into the trough 44 times. The resulting three-page profile, published in The New York Herald Tribune as part of its supplement This Week, ran under the headline “Make Way for A Lady” cq.
The article called her discovery ‘invisible glass”, which we know irked Katharine because it is not really accurate. She was right, of course. Glass itself is not invisible, her films just cut out the glare. Several years later, Katharine corrected a reporter from the Associated Press on exactly this point: “Dr. Blodgett frowns on the phrase invisible glass,” the piece read. She always says, "Non-reflecting.”
Optometrists descended on Schenectady to learn more about how Katharine’s discovery might reduce reflection from spectacle lenses. Their visit, of course, was written up in the local paper.
Let’s just call 1939 the year of Katharine Blodgett. Because on top of what I just mentioned here’s what else happened: she was awarded an honorary Doctor of Science degree at Elmira College in upstate New York and GE put out another press release about that). And oh, the world in 1939 was so very different from today, because when she was admitted to the American Physical Society, the letter informing her of the honor was addressed as “Dear Sir.”
In July of 1939, Katharine boarded the Queen Mary for a well-earned vacation in England, and to give a lecture or two on the side. Her many letters to her mother were positively ebullient as she chronicled time spent at her old haunts in Cambridge, lunch with Lady Darwin (yes, of that Darwin family), a talk at the National Physical Laboratory, a sojourn in London filled with theater, and a lecture in Manchester, in the north of England.
Back to the US, the invitations to speak kept pouring in. The media interest just kept on going. Katharine was named one of the “Most Distinguished Women of 1939” in the widely-read “American Women” Magazine, her name published alongside other prominent women, including the actress Bette Davis, socialite Elsa Maxwell, and First Lady Eleanor Roosevelt.
Even Irving Langmuir joined in on the praise of his associate, which no doubt pleased her immensely. According to a G.E. press release, in March of 1940, he spoke at a dinner hosted by the Schenectady Advertising Club in Katharine’s honor. He declared that “a woman can be as good a scientist as a man” – high praise indeed! – and he called Katharine “a gifted experimenter, "a good mathematician," and a woman who has “that rare combination of theoretical and practical ability." At that same dinner, a personalized note from none other Mrs. Roosevelt was read aloud.
Most articles covered Katharine’s scientific prowess and her discovery, though the writers also wanted to know about Katharine the woman, her pastimes, her gardening and her cooking. As usual, she was patient and obliging.
The San Francisco Chronicle described her thusly: “Five feet, one and a half inches tall and weighing 115 pounds, she epitomizes the theory of perpetual motion.” Okay, that’s annoying. It’s hard to believe a reporter asking how much a male scientist weighed!
This recognition and adulation came at the end of a decade that was perhaps the hardest in Katharine’s working life. Yes, it brought great success and a measure of fleeting fame, and yes, she conducted her work patiently and intensely. Her co-workers never imagined what else might be going on.
Here’s Vincent Schaefer, again:
Vincent Schaefer: I never saw Katie depressed. Sure. There were times when her popovers weren't working as well as she expected they would, or when something went wrong in her garden or some other thing happened, which took a little of the gloss off.
But unfailingly, she had this enthusiastic approach to things, which was a joy to see.
Katie Hafner: Vincent had no idea how wrong he was.
NEXT WEEK ON Layers of Brilliance
Katie Hafner: There was a lot more to the Katharine people thought they knew... there was a Katharine, who, in fact, almost no one knew...
Deborah Alkema: Um, I need to get in my storage unit around there. This one here.
Katie Hafner: Oh, wow.
Deborah Alkema: We have a lot of family papers.
Katie Hafner: This has been Lost Women of Science. The producers of this episode were Natalia Sánchez Loayza and Sophia Levin, with me as senior producer. Hannah Sammut was our associate producer. Elah Feder was our consulting editor. Ana Tuirán was our sound designer and Hansdale Hsu was our sound engineer.
Elizabeth Younan is our composer and Lisk Feng designed the art.
Thanks to senior managing producer Deborah Unger, program manager Eowyn Burtner, my co-executive producer Amy Scharf, and marketing director Lily Whear.
We got help along the way from Eva McCullough, Nadia Knoblauch, Theresa Cullen, and Issa Block Kwong.
A super special thanks to Peggy Schott. Special thanks as well to Michael Petty and Osvaldo Oliveira.
And we're grateful to Deborah, Jonathan, and Marijke Alkema for helping us tell the story of their great Aunt Katharine. We're distributed by PRX and our publishing partner is Scientific American. Our funding comes in part from the Alfred P Sloan Foundation and the Anne Wojcicki Foundation, and our generous individual donors
Please visit us at lost women of science.org, and don't forget to click on that all-important donate button. I'm Katie Hafner. See you next week.
Listen to the Next Episode in this Series
More Episodes
Listen to the complete collection of episodes in this series.